See how HealthScribe protects →
What are the steps HealthScribe follows?
Step 01
Step 02
Step 03
Step 04
Step 05
Watch a sample intake flow →
Last 30 Days
Q3 Customer Support
2 variants • 45k runs
Sales Outreach Bot
1 variant • 12k runs
Governance line: Your team stays in control: HealthScribe suggests what to look at, and people decide what to change and approve.
Systems
Connects to your CTMS, eIRB, and document systems to pull the latest versions of protocols and consents.
Works alongside Word, Google Docs, sponsor templates, and site systems, so your team feels supported without workflow disruption.
Sends status, reports, and alignment notes back into your existing systems so teams can see them in their normal workflows.
Who uses it
Medical writers and clinical scientists who draft protocols and consents.
Regulatory affairs and IRB liaisons who prepare submissions and handle “modifications required.”
Principal investigators and research leaders who sign off on the final content.
Site staff who rely on clear, consistent documents to explain studies to participants.
Faster start-up and reconsent
Teams skip manual comparisons, speeding submissions and easing workload.
Fewer IRB comments and delays
Catch mismatches or discrepancies early to avoid rework and review delays.
Clear proof for audits
Every change is logged for easy traceability with sponsors and regulators.
Less burnout for experts
Experts focuses on critical decisions, not minor wording checks.
Less site and participant burden
Sites avoid backandforth, and participants get clear, consistent study info.
Governance Mini block
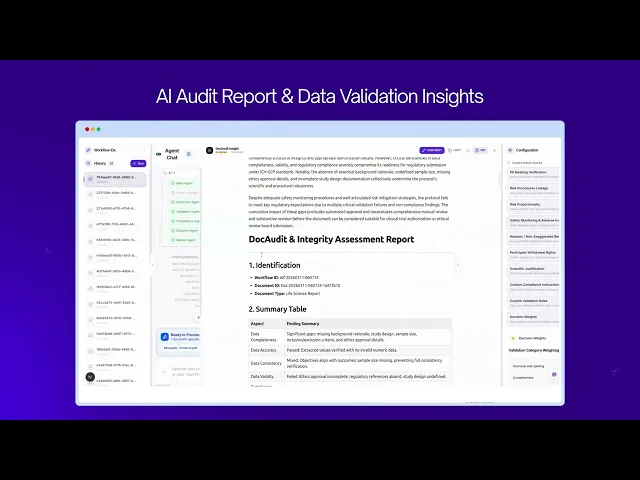
HealthScribe helps regulated environments by providing full logging of checkpoints, issue identification, and how they were resolved; all governed by your own rules and review thresholds.
See examples of reports and dashboards →
Establishes the approved study protocol (or amendment) as the single source of truth.
Automatically validates every consent form, flyer, web page, and handout against the latest protocol, no manual checks needed.
Operates flexibly across modes: live in Word or Google Docs, in bulk from your CTMS, or on a schedule after each amendment.
Questions teams ask before they turn PA agentic
Ready to see where your documents don’t match before the IRB does?
